Density Investigations
By E Saylor
Question #1
- An older penny, minted before 1982, is made of pure copper. If the density of copper is 8.9 g/cm3, what is the mass of the penny if its volume is 0.35 cm3.
- Using a triple beam balance, find the mass of a copper penny. Using this as the experimental value, find the % error between your measured value and the theoretical value from part A.
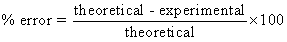
Question #2
- A newer penny, minted after 1982, is mostly zinc. If the volume of a penny is 0.35 cm3 and the density of zinc is 7.14 g/cm3, what is the theoretical mass of the penny?
- Using a triple beam balance, find the mass of a zinc penny. Using this as the experimental value, find the % error between your measured value and the theoretical value from part A.
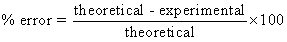
Question #3
- If a block of ice (density = 0.917 g/cm3) measures 10.0 cm by 5.0 cm by 7.5 cm, what will its mass be? Remember that the volume of a rectangular solid is length x width x height.
- The block of ice is cut in half. Does its density change? Explain.
- Does ice (density = 0.917 g/cm3) sink or float in water (density = 1.00 g/cm3)?
- Using a syringe (to measure volume) and a triple beam balance (to measure mass), find the experimental density of water.
- If the theoretical density of water is 1.0 g/cm3, what is the % error?
Question #4
- Lead is a soft metal that has high density. Find the density of the piece of lead at your table. You can find the volume of the lead using water displacement.
- If the theoretical value for the density of lead is 11.4 g/cm3, how much error is there between your experimental value and the theoretical value?
- Explain, using density, why lead is used to make sinkers for fishing.
Question #5
Gasoline (density = 0.67 g/cm3) will not mix with water. Draw a diagram showing gasoline and water. Label each liquid.