Mass of Pennium: Average Atomic Mass of Isotopes
By E Saylor
Problem: What is the mass of pennium?
Materials:
Old and new penniesTriple beam balance
Procedure:
- Count and record the number of each kind of penny: old and new.
(isotope A) # of old pennies = _____
(isotope B) # of new pennies = _____
- Calculate the percentage of occurrence of each isotope.
- Find the mass of one old penny and the mass of one new penny.
Mass of isotope A = _____ g
Mass of isotope B = _____ g
- Calculate the average atomic mass of pennium.
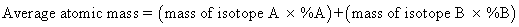
This will be the experimental value for the mass of pennium.
- Now, calculate the average atomic mass by massing ALL the pennies and dividing by the total # of pennies.
This will be the theoretical value for the mass of pennium.
- Calculate the % error.