Word Problem Exercises: Science - Density Problems
By G Waller
What is the density of a piece of wood that has a mass of 25.0 grams and a volume of 29.4 cm3?
A piece of wood that measures 3.0 cm by 6.0 cm by 4.0 cm has a mass of 80.0 grams. What is the density of the wood? Would the piece of wood float in water? (volume = L x W x H)
Gold: 19.3 g/mLCopper: 8.86 g/mL
Bronze: 9.87 g/mL
I threw a plastic ball in the pool for my dog to fetch. The mass of the ball was 125 grams. What must the volume be to have a density of 0.500 g/mL. ( I want it to float of course!)
After throwing the ball in the pool for my dog, the ball sprung a leak and began to fill with water. How many mL of water can the ball absorb before the ball sinks?
What is the mass of a cylinder of lead that is 2.50 cm in diameter, and 5.50 cm long. The density of lead is 11.4 g/mL and the volume of a cylinder is 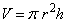 .
.
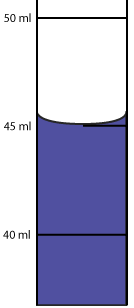
An ice cube measuring 5.80 cm by 5.80 cm by 5.80 cm has a density of 0.917 g/mL. What is the mass?
Gasoline is a non-polar liquid that will float on water. 450 grams of gasoline is spilled into a puddle of water. If the density of gasoline is 0.665 g/mL, what volume of gasoline is spilled?
The density of aluminum is 2.70 g/mL. If the mass of a piece of aluminum is 244 grams, what is the volume of the aluminum?
Directions and/or Common Information:
A Mind Bender ProblemA little aluminum boat (mass of 14.50 g) has a volume of 450.00 cm3. The boat is place in a small pool of water and carefully filled with pennies. If each penny has a mass of 2.50 g, how many pennies can be added to the boat before it sinks?