Chemistry Graphs: Progression of Reaction
By E Saylor
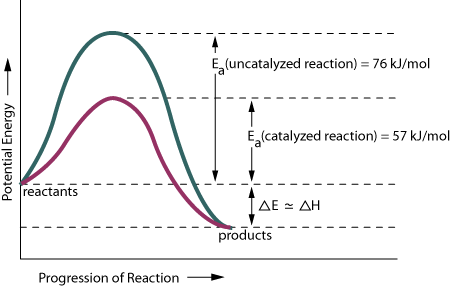
Use the given graph to answer the following questions.
According to the graph, for this particular reaction, the reactants have a (larger, smaller) amount of stored energy than the products.
The uncatalyzed reaction requires more energy to start up than does the catalyzed reaction. How much more energy is required when the catalyst is not present?
What does ΔH represent?
Some chemical reactions are reversible. If this reaction were reversed, would it be an exothermic or an endothermic process?
Does the graph more closely represent the melting of ice or the striking of a match?