Chemistry Graphs: Pressure, Temperature, and Equilibrium
By E Saylor
One mole of nitrogen gas combines with three moles of gaseous hydrogen to create two moles of gaseous ammonia according to the following equation:
N2(g) + 32(g) 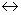 2NH3(g)
2NH3(g)
This reaction is commercially important in the production of fertilizer. By adjusting the pressure and temperature, the reaction can be pushed forward, favoring the production of ammonia. Since German chemist Fritz Haber was instrumental in formulating a method for synthesizing ammonia, the reaction is closely associated with his name.
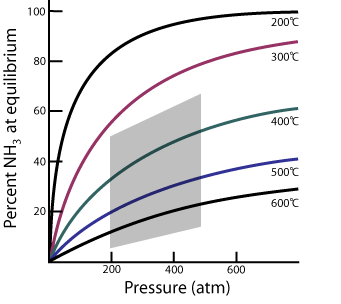
According to the graph, do high or low temperatures favor the formation of NH3?
If the percent of NH3 decreases at higher temperatures, is the reaction exothermic or endothermic?
According to the graph, what is the range of pressures and temperatures used in the Haber process?
From the information on the graph, what is the relationship between pressure and the percent of NH3 at equilibrium?
What is the percent of ammonia generated when production is done at 400oC and 400 atmospheres of pressure?