Chemistry Graphs: Potential Energy Diagrams
By E Saylor
Use the given graphs to answer the following questions.
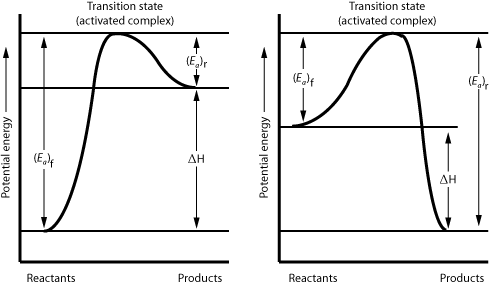 | |
A | B |
Activation energy is the energy that must be gained by the reactant molecules in order to initiate a reaction. Compare the activation energy shown on graph A to the activation energy on graph B. Which reaction is more favorable?
Which reaction is endothermic?
The highest point on the graph represents the activated complex. If the complex loses energy, what will it transform into?
Some reactions are reversible. In order to decide which direction is favored, a comparison of the activation energy in each direction must be made. For graph A, which direction is favored, forward or reverse?
Potential energy is stored within the bonds of molecules. For an exothermic reaction, such as that plotted on graph b, do the reactant molecules or product molecules possess a greater amount of stored energy?