Chemistry Graphs: Density
By M Aumiller
Density is defined as the ratio of the mass of a given sample of a substance (M) to its volume (V). It is usually measured in units such as kg/m3, g/cm3, or g/mL.
Use the following graph of Volume vs. Mass to answer the next three questions.
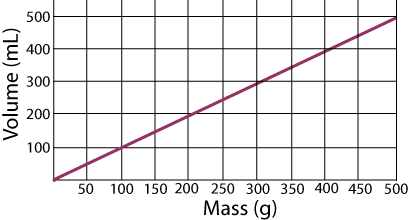
As the volume of a sample increases from 200. mL to 300. mL, does the mass increase or decrease?
According to this graph, what is the density of the substance being studied?
Based on the information provided in the chart below, what substance was represented by the data displayed on the graph? The densities are measured in g/mL and the small letters represent the state of the substance at room temperature: solid, liquid, or gas.
Substance | Density |
Hydrogen(g) | 0.00009 |
Mercury(l) | 13.5 |
carbon(s) | 2.3 |
ammonia(g) | 0.008 |
water(l) | 1.00 |
sucrose(s) | 1.6 |