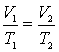Chemistry Graphs: Charles' Law
By M Aumiller
Charles' Law states that the volume (V) of a confined gas is directly proportional to its temperature (T) in Kelvin.
When the temperature changes one unit on the Kelvin scale it is equivalent to a change in one Celsius degree; that is, they are in a one-to-one correspondence. Note that 0 on the Kevin scale represents "Absolute Zero" or -273 ºC.
Use the following graph of Volume vs. Temperature to answer the next four questions.
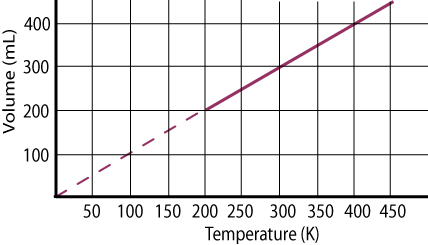
As the temperature of the gas increases from 250 K to 350 K, does the volume of the gas increase or decrease?
What is the volume of this gas sample when its temperature is 300 K?
What is the temperature of the gas when it occupies a volume of 250 mL?
If 0.05 moles of gas are present in our sample, use the slope of the line to determine the constant pressure that was exerted on the gas during this experiment.