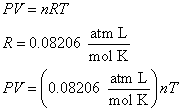Chemistry Graphs: Boyle's Law
By M Aumiller
Boyle's Law states that the volume of a confined gas is inversely proportional to the pressure exerted upon it.
Use the following graph of Volume vs. Pressure to answer the next four questions.
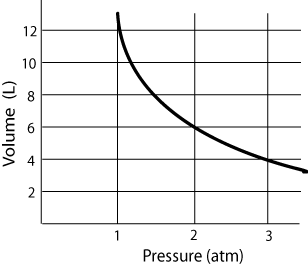
At a pressure of 3 atmospheres, what is it the volume of the gas?
As the pressure of the gas increases from 1 atmosphere to 2 atmospheres, does the volume increase of decrease?
When the volume of the gas is 8 L, what is pressure is being exerted upon it?
Using your values for pressure and volume from question 3, along with the fact that room temperature is 298 K (25oC), calculate the number of moles of the gas using the Ideal Gas Law.