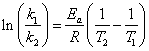Chemistry Graphs: Arrhenius Plot
By E Saylor
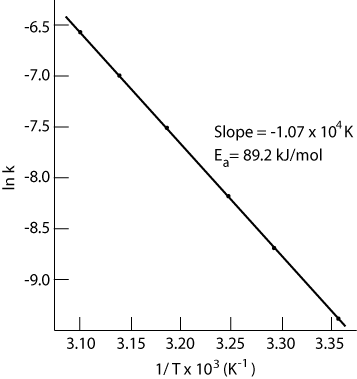
The rate constant, k, for a given reaction changes with temperature. The plot of ln k versus 1/T gives a linear plot with a negative slope. Ea represents the activation energy for the reaction, in KJ/mole. R is the ideal gas law constant, 8.314 J/mole K. Temperature is measured in Kelvin.
The equation of the line for the graph is:
If the slope of the line is -1.10 x 103 K, what is the activation energy for the reaction?
If ln k = -7.1, what is the value of k?
Will the rate constant, k, increase or decrease as the temperature increases?
What is the unit for the variable being plotted on the x-axis?
Simplify the following expression: ln k1 - ln k2