Science: Electromagnetic Radiation - Wavelength, Frequency, and Photon Energy
By E Saylor
Useful Information: Note that both c and h are constants.
c = speed of light = 3 x 1010 cm/sec = 3 x 108 m/sech = Planck's constant = 6.626 x 10-34 J/sec
Useful Formulas
c = wavelength x frequency
energy of a photon = h x frequency
Useful Units and Conversions
nano means "billionth": 1 nm = 1 x 10-9 m OR 1 x 109 nm = 1 mmega means "million": 1 x 106 Hz = 1 MHzFrequency refers to the number of waves that pass by each second and is measured in a unit called Hertz (Hz).
Directions: Find the requested information in each question by using the formulas provided above.
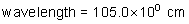
frequency: ?
energy of a photon: ?
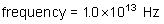
wavelength: ?
energy of a photon: ?
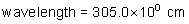
frequency: ?
energy of a photon: ?
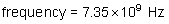
wavelength: ?
energy of a photon: ?
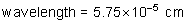
frequency: ?
energy of a photon: ?
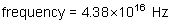
Wavelength: ?
Energy of a photon: ?
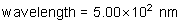
frequency: ?
energy of a photon: ?
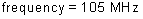
wavelength: ?
energy of a photon: ?
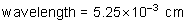
frequency: ?
energy of a photon: ?
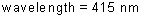
frequency: ?
energy of a photon: ?
Electromagnetic radiation propagates through space as waves. All graphs represent the number of waves propagating through the same distance in a one-second interval. Remember that all forms of electromagnetic radiation travel at the speed of light through a vacuum.
Which of the following waves would have the lowest energy photons? In answering this question, be sure to compare all three waves in terms of their relative wavelength, frequency, as well as energy.