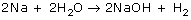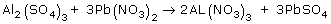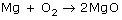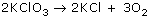Chemistry: Mole-Mole Ratios
By E Saylor
The coefficients (the numbers that precede a chemical formula) in a balanced equation indicate the mole ratio.
Example: A reaction uses 10.0 moles of O2. Predict how many moles of KClO3 will react.
Solution: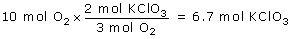
A reaction uses 15.0 moles of MgO. Predict how many moles of O2 will react.
A reaction uses 4.00 moles of NaOH. Predict how many moles of H2O will react.
A reaction uses 150. moles of PbSO4. Predict how many moles of Al2(SO4)3 will react.
A reaction uses 0.250 moles of KCl. Predict how many moles of O2 will react.
A reaction uses 9.00 moles of Pb(NO3)2. Predict how many moles of Al(NO3)3 will react.