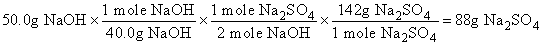Chemistry: Stoichiometry
By E Saylor
2 NaOH + H2SO4 --> 2 H2O + Na2SO4 | |
Compound | Molar Masses (g/mole) |
NaOH | 40.0 |
H2SO4 | 98.1 |
H2O | 18.0 |
Na2SO4 | 142 |
Example:
If the reaction starts with 50.0g of NaOH, how many grams of Na2SO4 will form?
Solution:
If the reaction consumes 100 g of NaOH, how many grams of Na2SO4 should form?
If 475 g of H2O form, predict how many grams of NaOH must have been consumed?
The reaction generates 35.0 g of Na2SO4. How many grams of H2SO4 should have been used up?
If 25.0 g of NaOH is used, how many grams of H2SO4 should react?
Directions and/or Common Information:
A reaction produces 100 g of H2O.How many grams of NaOH will be consumed?
How many grams of H2SO4 will be consumed?
If the law of conservation of mass holds true, the total mass of the reactants (NaOH and H2SO4) should equal the total mass of the products (H2O and Na2SO4). Without doing a stoichiometry problem, predict how many grams of Na2SO4 should form.