Specific Heat
Subject undefined
Pronunciation: spe•CIF•ic HEAT
Definition
the amount of heat (measured in Joules or calories) required to raise the temperature of 1 kilogram of a substance 1 Celsius degree; c = Q / (mΔT)
Example
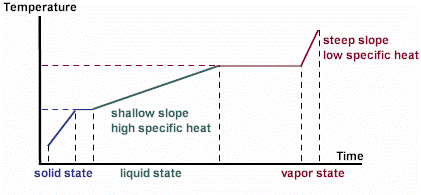
On the following diagram a constant amount of heat is being added each second to a given sample. Notice that the amount of heat required to raise the temperature of the substance when it is in its liquid state is much greater (since it takes more time) than the amount of heat required to raise its temperature when it is in its vapor state.