First Law of Thermodynamics
Subject undefined
Pronunciation: FIRST LAW OF ther•mo•dy•NAM•ics
Definition
when heat is added to a closed thermal system either the internal energy of the system increases or the volume of the system changes reflecting that work has been done
Example
During an isothermal expansion, a confined gas doubles it volume. During this process, heat must be added to the system in order to keep the temperature of the gas constant. The amount of heat added exactly matches the work done by the gas.
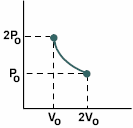 |
|